Research findings related to UNITE work package A01
Purpose: The EORTC-26101 study was a randomized phase II and III clinical trial of bevacizumab in combination with lomustine versus lomustine alone in progressive glioblastoma. Other than for progression-free survival (PFS), there was no benefit from addition of bevacizumab for overall survival (OS). However, molecular data allow for the rare opportunity to assess prognostic biomarkers from primary surgery for their impact in progressive glioblastoma.
Experimental design: We analyzed DNA methylation array data and panel sequencing from 170 genes of 380 tumor samples of the EORTC-26101 study. These patients were comparable with the overall study cohort in regard to baseline characteristics, study treatment, and survival.
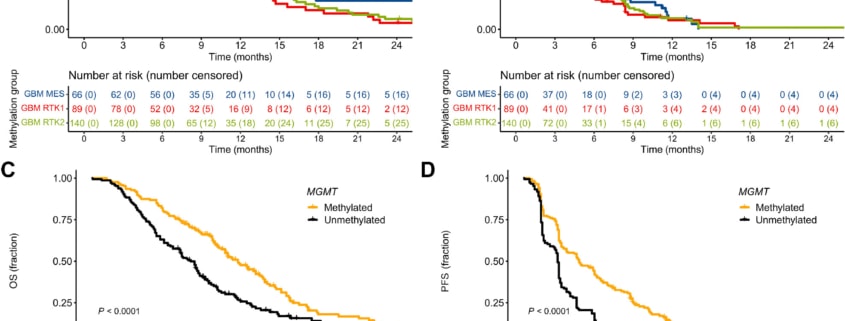
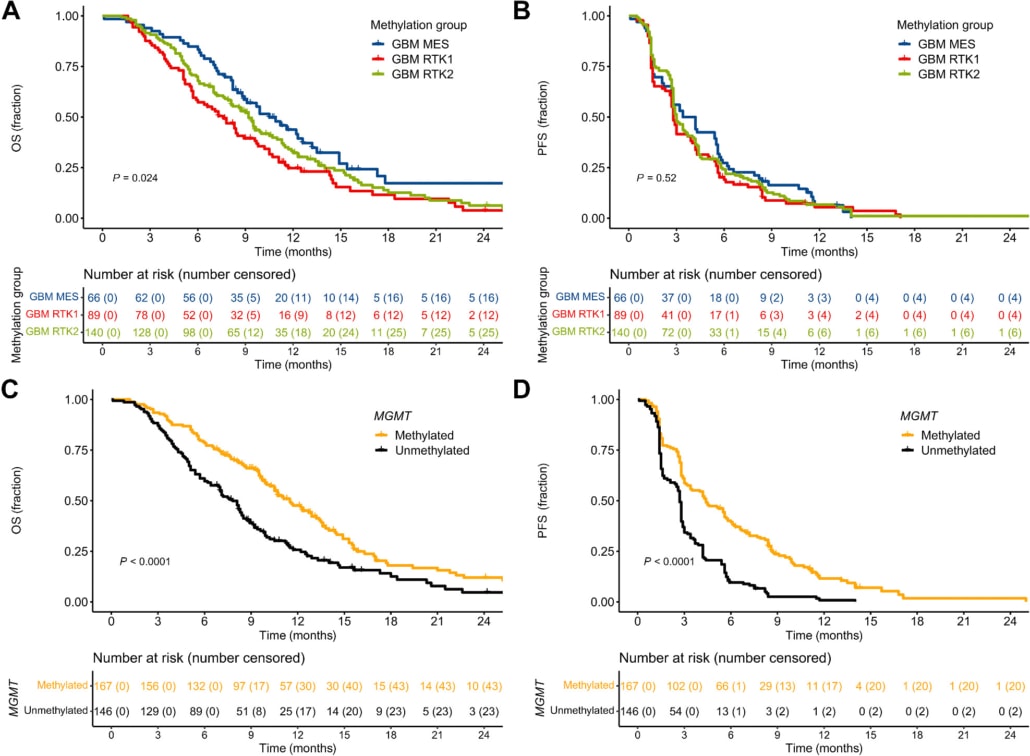
Results: Of patients’ samples, 295/380 (78%) were classified into one of the main glioblastoma groups, receptor tyrosine kinase (RTK)1, RTK2 and mesenchymal. There were 10 patients (2.6%) with isocitrate dehydrogenase mutant tumors in the biomarker cohort. Patients with RTK1 and RTK2 classified tumors had lower median OS compared with mesenchymal (7.6 vs. 9.2 vs. 10.5 months). O6-methylguanine DNA-methyltransferase (MGMT) promoter methylation was prognostic for PFS and OS. Neurofibromin (NF)1 mutations were predictive of response to bevacizumab treatment.
Conclusions: Thorough molecular classification is important for brain tumor clinical trial inclusion and evaluation. MGMT promoter methylation and RTK1 classifier assignment were prognostic in progressive glioblastoma. NF1 mutation may be a predictive biomarker for bevacizumab treatment.
Kessler T*, Schrimpf D, Doerner L, Hai L*, Kaulen LD, Ito J, van den Bent M, Taphoorn M, Brandes AA, Idbaih A, Dômont J, Clement PM, Campone M, Bendszus M*, von Deimling A*, Sahm F*, Platten M*, Wick W*, Wick A. Prognostic Markers of DNA Methylation and Next-Generation Sequencing in Progressive Glioblastoma from the EORTC-26101 Trial. Clin Cancer Res. 2023 Jul 26; CCR-23-0926. doi: 10.1158/1078-0432.CCR-23-0926. *UNITE Principle Investigators

 UNITE
UNITE